Draw An Atom Of Carbon
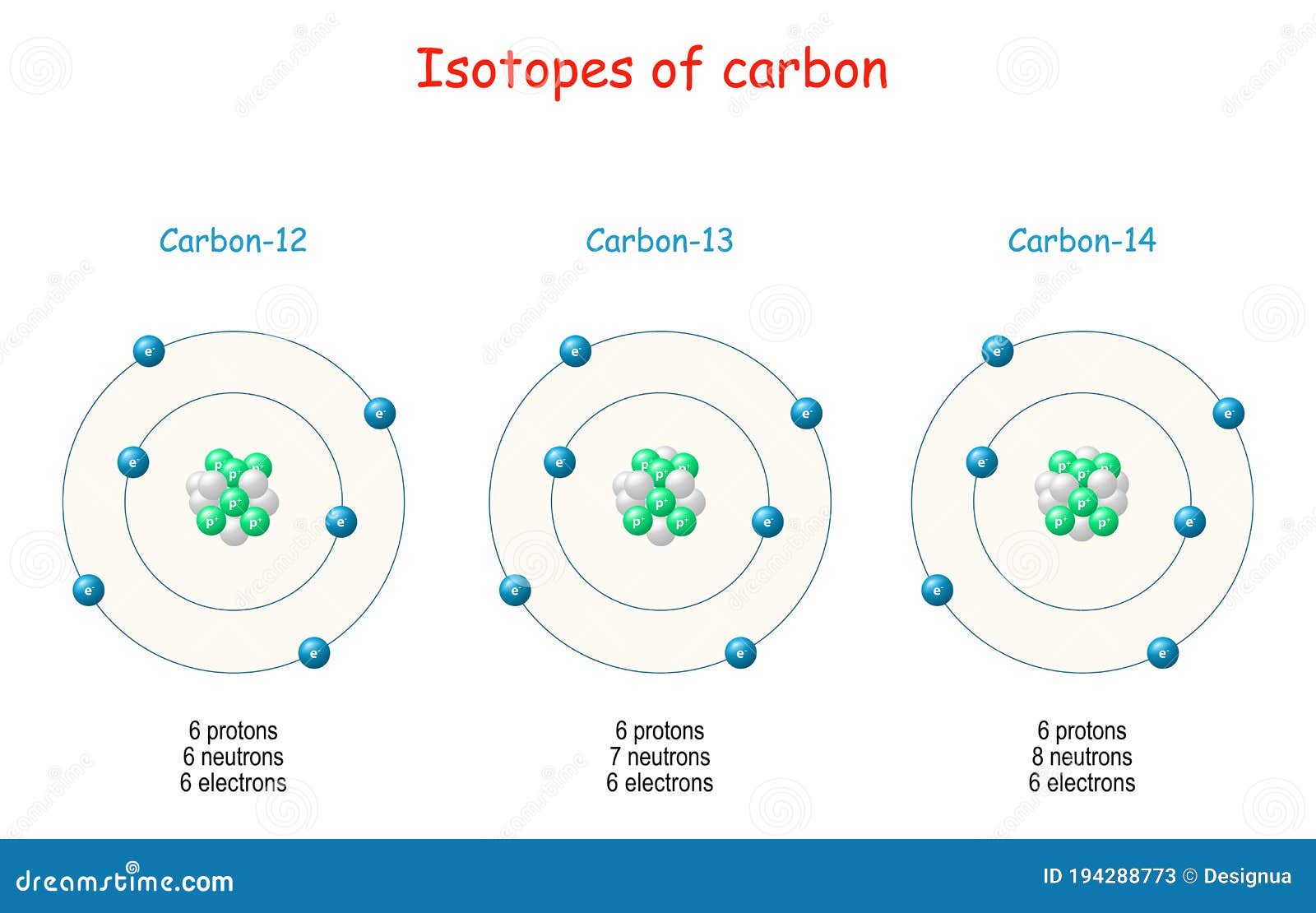
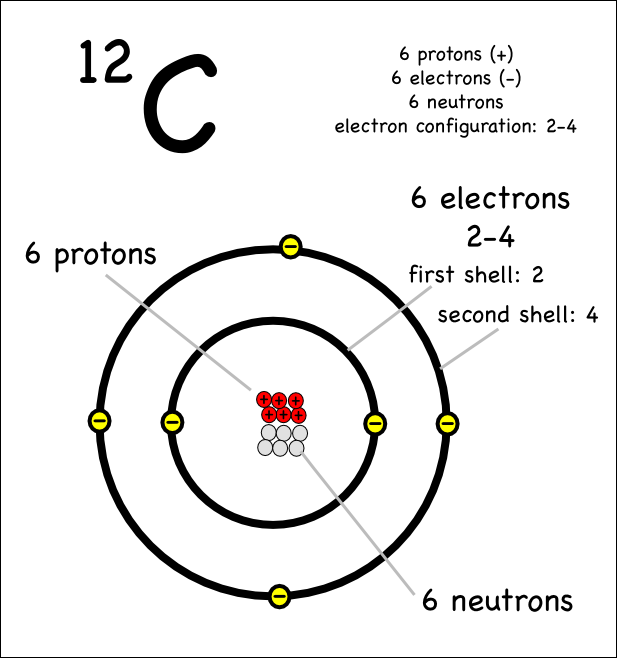
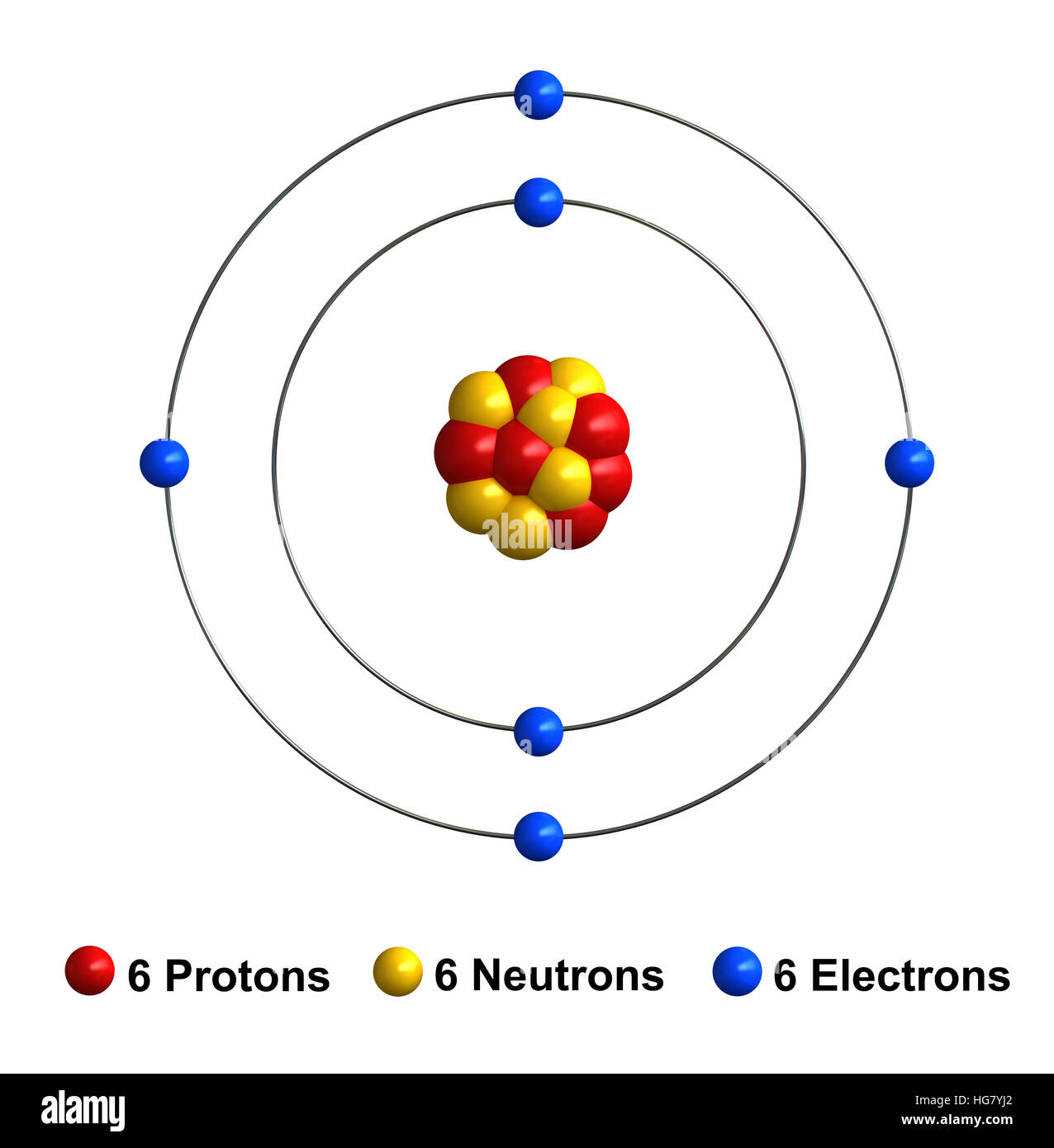
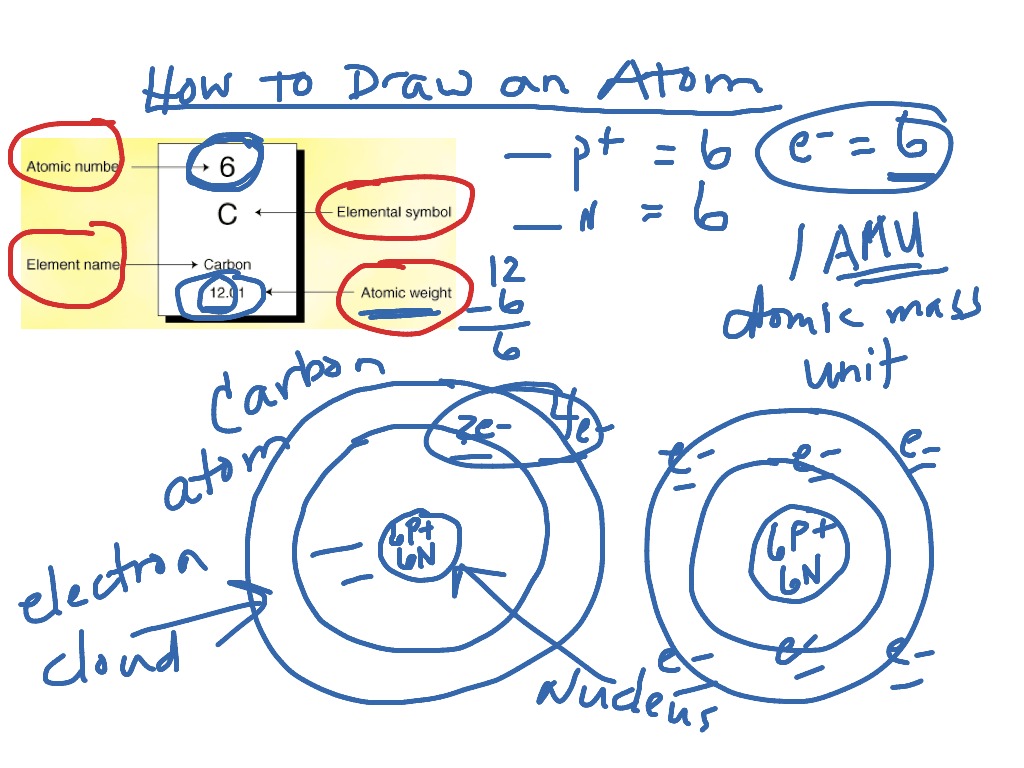
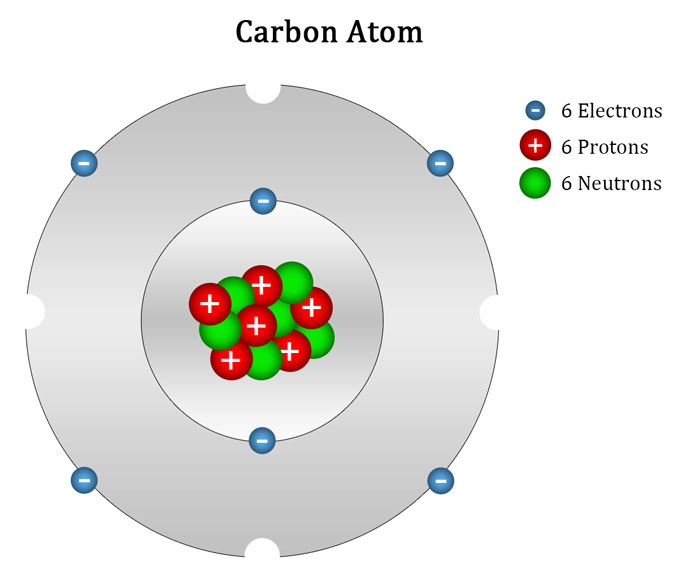
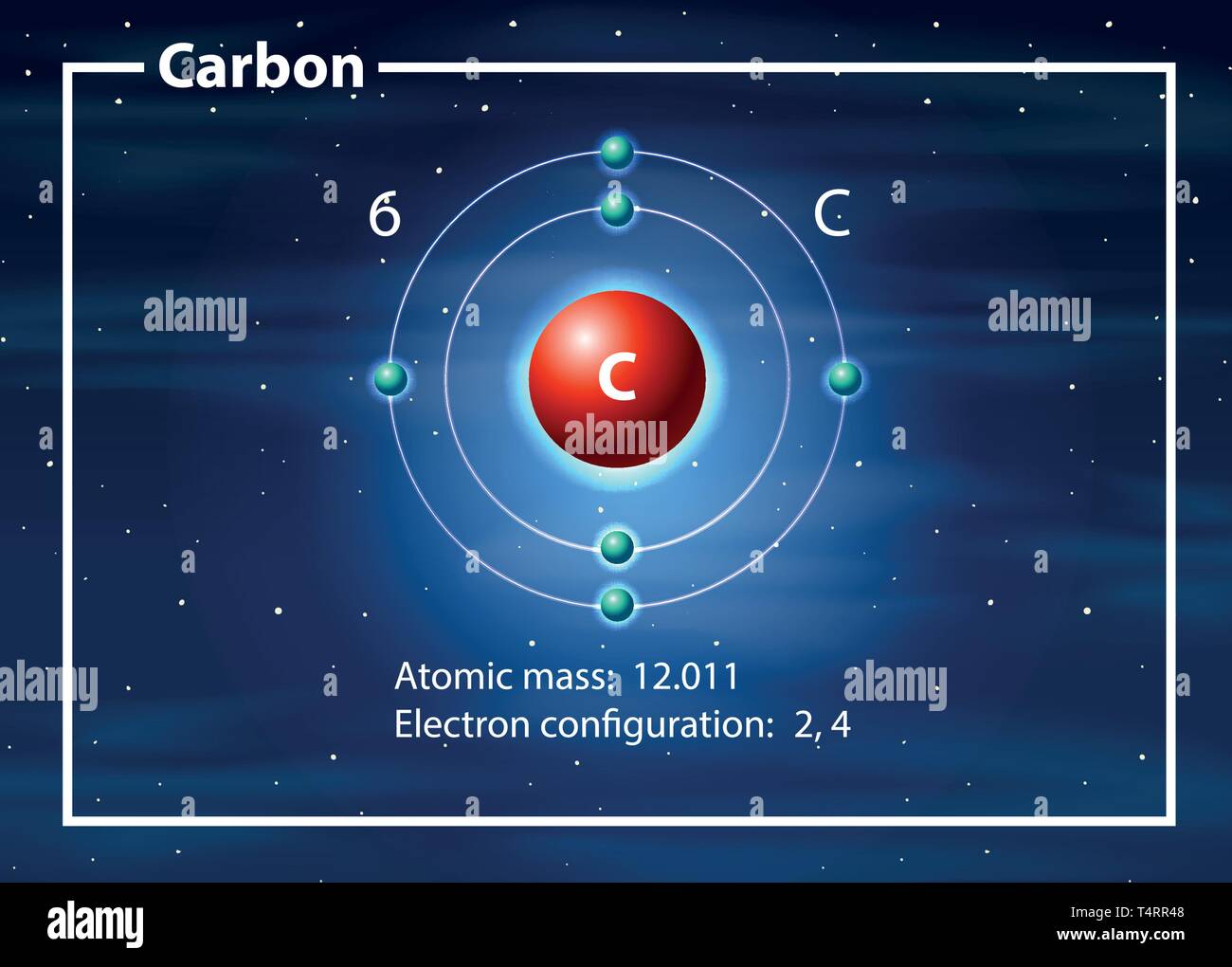
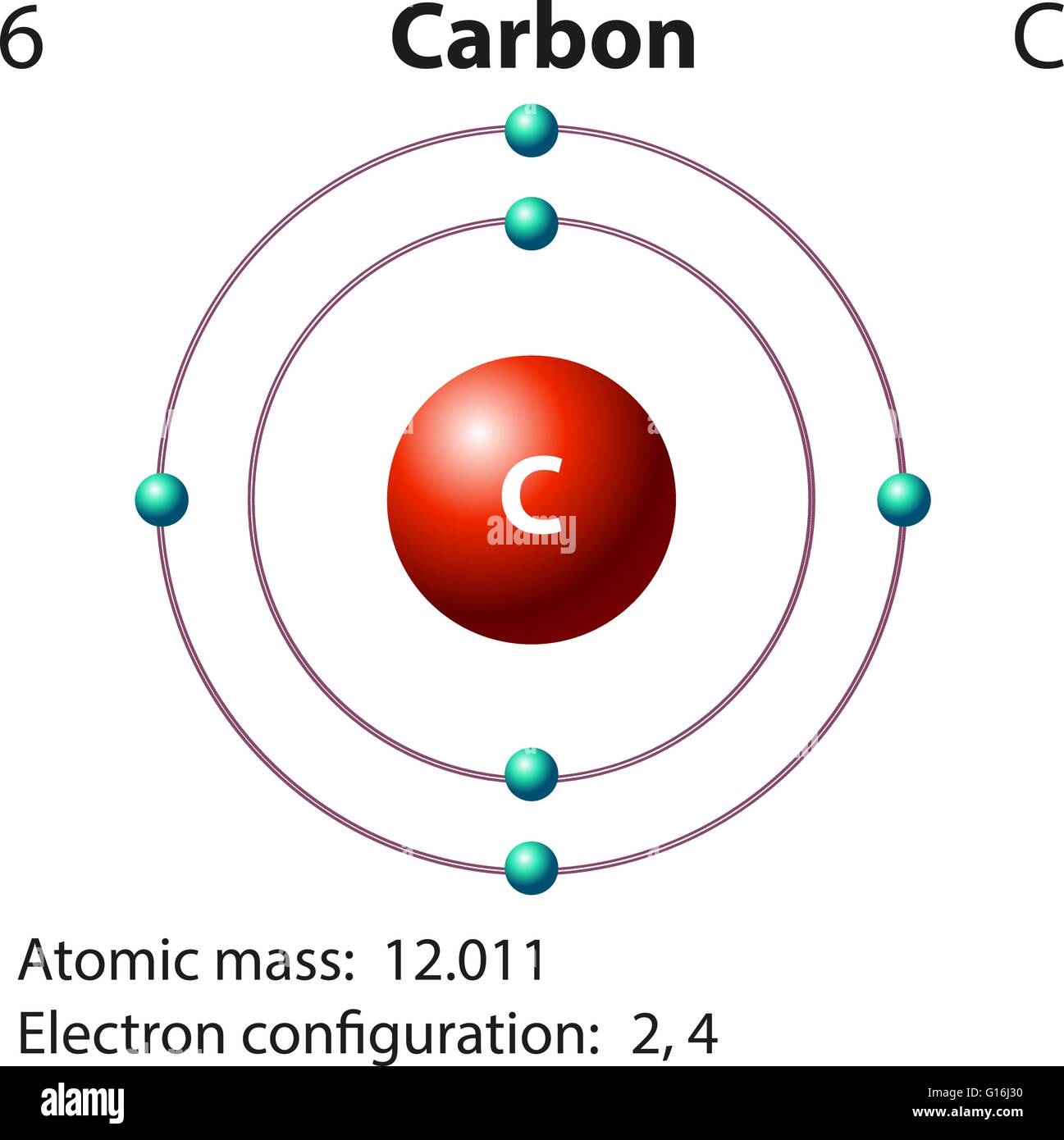
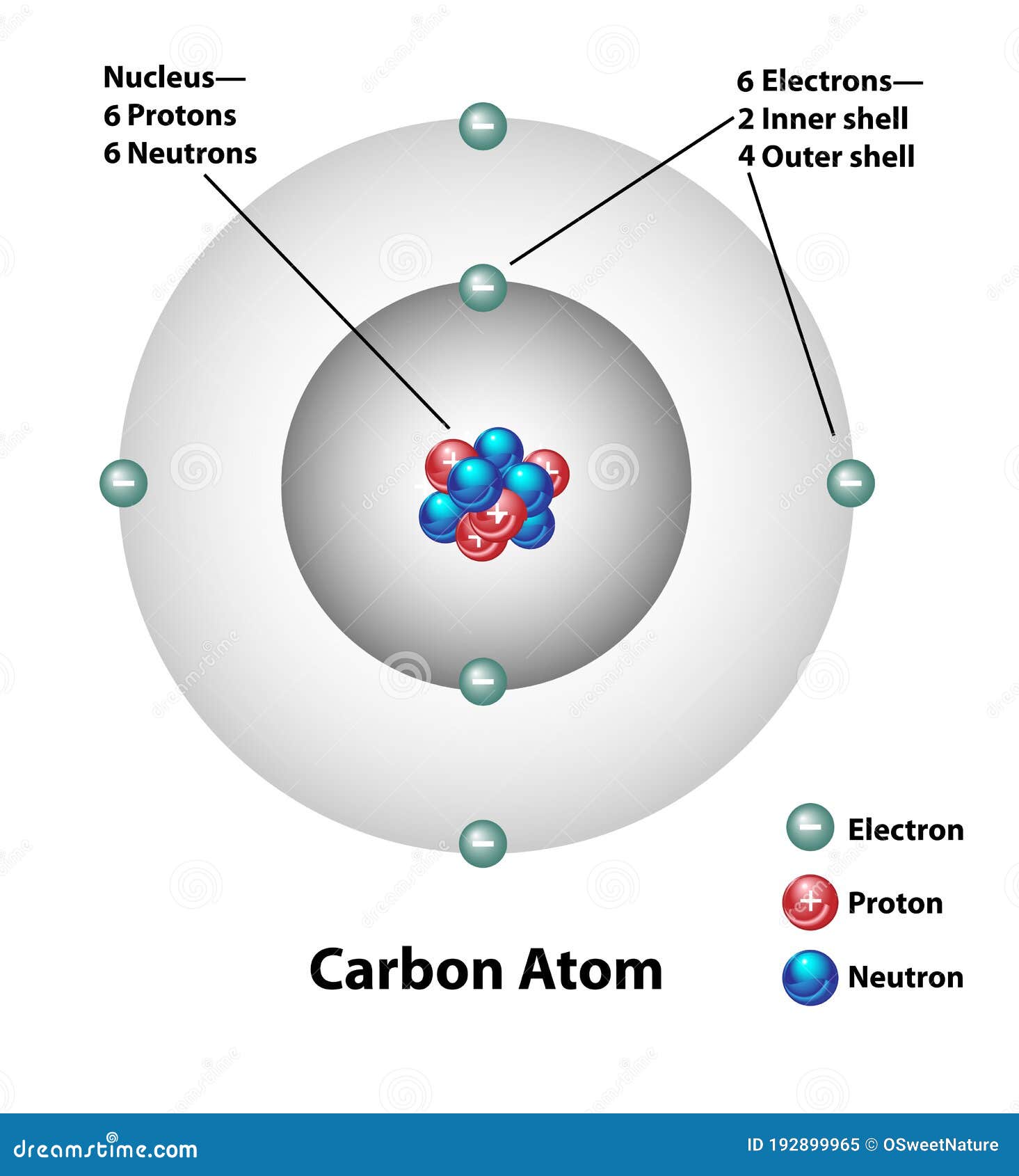
Draw An Atom Of Carbon - ⇒ the number of protons in a carbon atom = 6. Its electron configuration is 1s^22s^22p^2. Web the atomic number of carbon is 6, which is also the number of positively charged protons its atomic nuclei. Web carbon (from latin carbo 'coal') is a chemical element; Co 2 molecular geometry is based on a linear arrangement. Web since the overall formal charge is zero, the above lewis structure of co 2 is most appropriate, reliable, and stable in nature. The methyl group carbon atom has six valence electrons from its bonds to the hydrogen atoms because carbon is more electronegative than hydrogen. Web ⇒ the number of electrons in a carbon atom = 6. Web if the central atom has 8 or more electrons, you’re done. Since protons are the same as the amount of electrons, you just draw 6 protons. Your solution’s ready to go! So we're going to go ahead and put carbon in the center. ⇒ the number of neutrons in a carbon atom = 6. Neutrons are simply equal to the atomic mass minus the number of protons. Since carbon has an unpaired electron on all four sides of the electron dot structure that is shown above,. Web in a carbon atom, the nucleus, consisting of six protons and usually six neutrons, is surrounded by a cloud of electrons. Draw the nucleus of an atom. Erase the c in the center circle, and draw in your protons. Co 2 molecular geometry is based on a linear arrangement. A picture of a hydrogen atom can be found here. It has symbol c and atomic number 6. Just like protons, the number of electrons is also equal to the atom’s atomic number. First we determine the number of electrons in the atom; Web in cho 2 −, cho 2 −, the less electronegative carbon atom occupies the central position with the oxygen and hydrogen atoms surrounding it. It doesn't. Since carbon has an unpaired electron on all four sides of the electron dot structure that is shown above,. Web now, we will move forward to draw the shells of the carbon atom. And we know that the carbon is going to be bonded to an oxygen here. Web in cho 2 −, cho 2 −, the less electronegative carbon. As the energy level increases, the orbitals become larger and more complex. And we know that the carbon's going to be bonded to two hydrogens. Neutrons are simply equal to the atomic mass minus the number of protons. Therefore, the number of electrons in the carbon atom = 6. Web carbon has 2 electrons in its first shell and 4. It is possible to draw a structure with a double bond between a boron atom and a fluorine atom in bf 3, satisfying the. To draw the carbon bohr model, outline the 6 protons, 6 neutrons, and 6 electrons. For this, we would require calculating the number of electrons in this atom. Since carbon has an unpaired electron on all. We construct the periodic table by following the aufbau principle (from german, meaning “building up”). Web draw a valid electron dot structure for each of the given elements. Neutrons are simply equal to the atomic mass minus the number of protons. Web in a carbon atom, the nucleus, consisting of six protons and usually six neutrons, is surrounded by a. Erase the c in the center circle, and draw in your protons. Web in a carbon atom, the nucleus, consisting of six protons and usually six neutrons, is surrounded by a cloud of electrons. And we know that the carbon is going to be bonded to an oxygen here. Web if the central atom has 8 or more electrons, you’re. It is possible to draw a structure with a double bond between a boron atom and a fluorine atom in bf 3, satisfying the. Co 2 molecular geometry is based on a linear arrangement. The orbital diagram shows how the electrons are arranged within each sublevel. The change is very slight. It belongs to group 14 of the periodic table. Energy 0 1 1 x i ? The presence of a sigma bond and valence electron pairs repelling each other force them to move to the opposite side of the. Web carbon (from latin carbo 'coal') is a chemical element; Web oxygen is more electronegative, therefore carbon is going to be at the center of our dot structure. Web draw. The bohr model shows the atom as a central nucleus containing protons and neutrons, with the electrons in circular electron shells at specific distances from the nucleus, similar to planets orbiting around the sun. Web since the overall formal charge is zero, the above lewis structure of co 2 is most appropriate, reliable, and stable in nature. Web the carbon. Noel pauller hope this helps! A nucleus is a dense and small region that contains the number of protons and. Web once you can draw a lewis symbol for an atom, you can use the knowledge of lewis symbols to create lewis structures for molecules. Draw the nucleus of an atom. Since protons are the same as the amount of. Web lewis dot diagram of carbon this video shows how to use the periodic table to draw lewis structures and figure out how many valence electrons an atom has. You could easily invent other ones as well. Web now, we will move forward to draw the shells of the carbon atom. And it does not satisfy the octet rule for the central be atom (see as follows). These are therefore two isotopes of carbon. First we determine the number of electrons in the atom; We construct the periodic table by following the aufbau principle (from german, meaning “building up”). So we're going to go ahead and put carbon in the center. In the full sketcher, deleting a bond will remove that bond and deleting an atom will remove that atom and any attached bonds. ⇒ the number of protons in a carbon atom = 6. It has symbol c and atomic number 6. Since carbon has an unpaired electron on all four sides of the electron dot structure that is shown above,. Its electron configuration is 1s^22s^22p^2. Web ⇒ the number of electrons in a carbon atom = 6. Carbon (c ), as a group. The length of the chain will be determined by the distance of the current draw position to the sprout.Carbon Atom Diagram
Drawing Atoms Montessori Muddle
Carbon Atomic Structure High Resolution Stock Photography and Images
How to draw an atom of carbon Science ShowMe
Carbon Atom Ascension Glossary
Carbon atom diagram hires stock photography and images Alamy
Carbon atom diagram concept Royalty Free Vector Image
Carbon atom Stock Vector Images Alamy
Carbon atomic structure (437243) Illustrations Design Bundles
Carbon Atom Molecular Structure Labels Stock Vector Illustration of
Web If The Central Atom Has 8 Or More Electrons, You’re Done.
Web In A Carbon Atom, The Nucleus, Consisting Of Six Protons And Usually Six Neutrons, Is Surrounded By A Cloud Of Electrons.
Surrounding This Nucleus Are Two Electron Shells, Containing A Total Of 6 Electrons.
Web Draw A Valid Electron Dot Structure For Each Of The Given Elements.
Related Post: